Total Phosphorous
How to interpret:
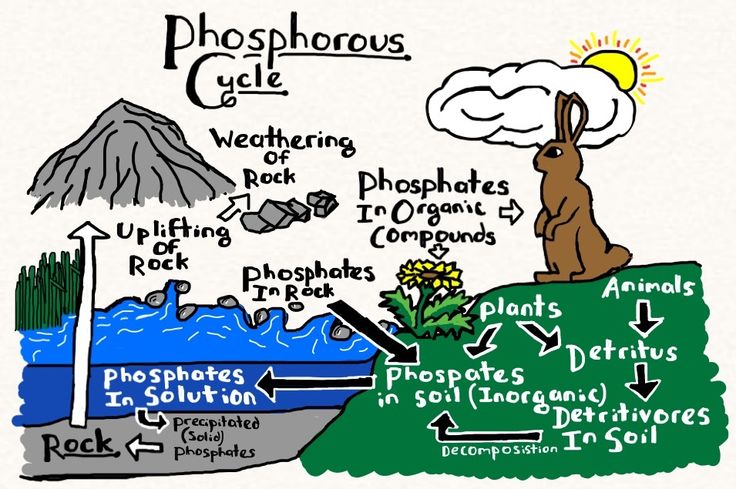
Total phosphorous measures the concentration of inorganic phosphate ions dissolved in a sample of water as well as the quantity of phosphorous contained in the suspended organic matter. Aquatic organisms are able to take it up quite efficiently and as a result concentrations in pristine water are generally quite low.
Typical phosphorous ranges for different aquatic communities:
- 1 - 3 ug/L - the level in uncontaminated lakes. Very little aquatic vegetation.
- 2.5 - 10 ug/L - level at which plant growth is stimulated. A nutrient-poor lake with clear water which supports some algae and phytoplankton.
- 10 - 20 ug/L - A lake which supports a moderate degree of vegetation.
- > 20 ug/L - accelerated growth and consequent problems: Likely a body of water with phosphorous input from outside sources.
What it means:
Phosphorous is the key nutrient for photosynthesis which is the most limited in aquatic environments. Phosphorous concentrations determine the quanity of plankton and algae a body of water can support; this degree of "primary productivity" through photosynthesis has enormous influence on the structure of ecological communities found in a body of water.