Dissolved Oxygen
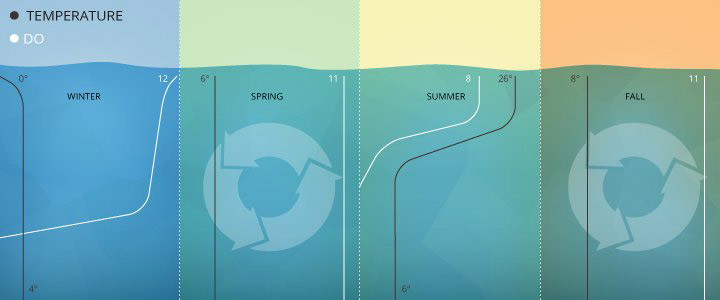
What is it? This is the amount of gaseous oxygen dissolved in the water. Oxygen gets into the water in various ways, including absorbing it from the atmosphere, by rapid movement of the water, or as a product of photosynthesis.
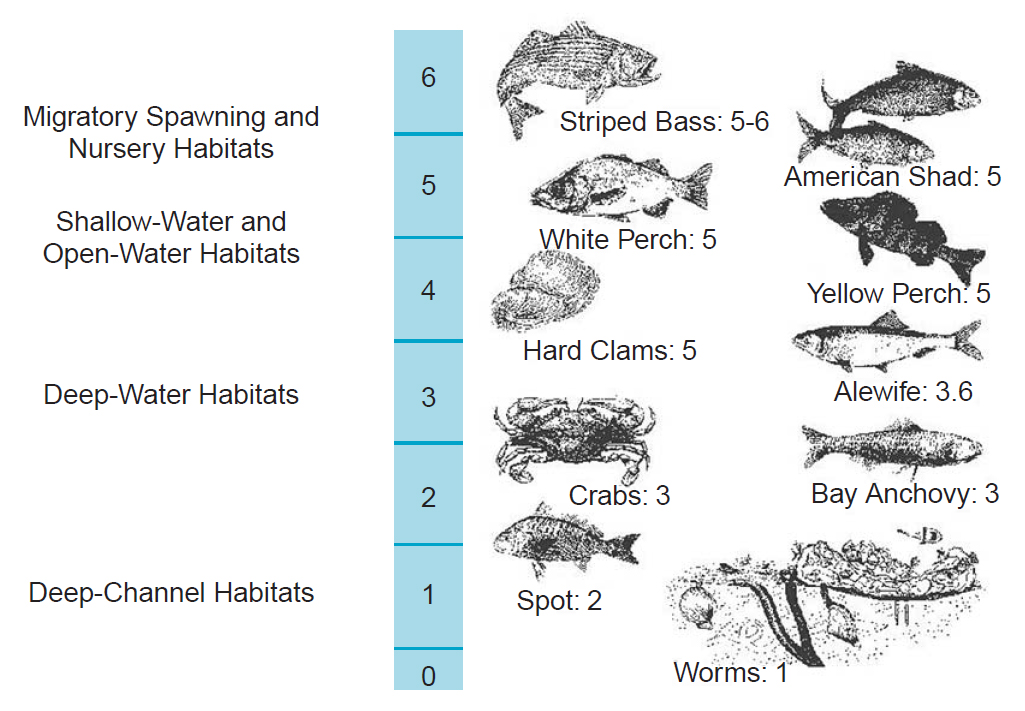
Why is it important? Oxygen is what gives the water life! Fish and plants alike depend on certain levels of oxygen to survive!
What does it mean? The percentage refers to the saturation level of oxygen in the water. The key factors in determining saturation are temperature, barometric pressure, altitude, and to a lesser extend salinity. For example, at sea level:
- 100% saturation at 25℃ = 8.26 mg/L
- 100% saturation at 0℃ = 14.6mg/L
Water Rangers Protocol: Since we do not have equipment for measuring barometric pressure and altitude this is not a standard measurement for us. However, we are working at building this into our app in the near future!